1.4 Electron Properties
2026-02-10 20:26
Status: #new
Tags: #chemistry #atoms
Electron charge relationships
Electronegativity
- How likely a particle becomes a [-] charge.
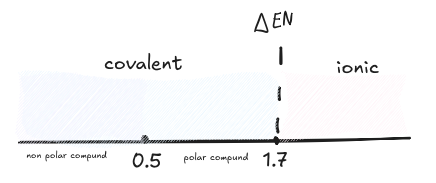
- This describes
values and what kind of bond they form. Note the polar cutoffs vary depending on professor as covalent ionic is a spectrum. - The periodic table trend is top right has highest EN.
Bonding Definitions
- Ionic Bond
- Attraction force between cations and anions
- Covalent bond
- Interaction between two atoms as they share
Bond Properties
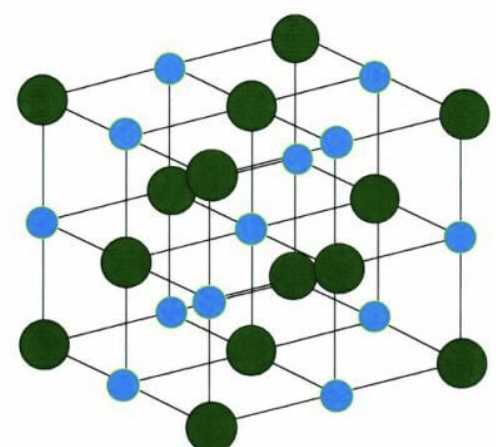
Crystalline Solvent
- An alternating pattern of cation anion atoms
Property Chart
| Property | Ionic | Covalent |
|---|---|---|
| State at room tempature | Crystal, Solid | Liquid, gas, solid |
| Melting point | High | Low |
| Electrical Conductivity | Yes | No |
| Solubility | Mostly high | Mostly low |
| Conductivity when dissolved | Yes | Not usually |
Formation process of ionic compound
- ie.
Electrical Conductivity
- A measure of the ability for an material to conduct electrical current
- Are there any charged particles inside said substance/material?
- Are they free moving?
- Solid Ionic compounds cannot conduct electricity